1. Introduction
2. Materials and Methods
2.1 Mortar composition
2.2 Incubation
2.3 Chlorophyll-a
2.4 Extracellular enzymes
2.5 Compressive strength
2.6 Statistical analysis
3. Results and Discussion
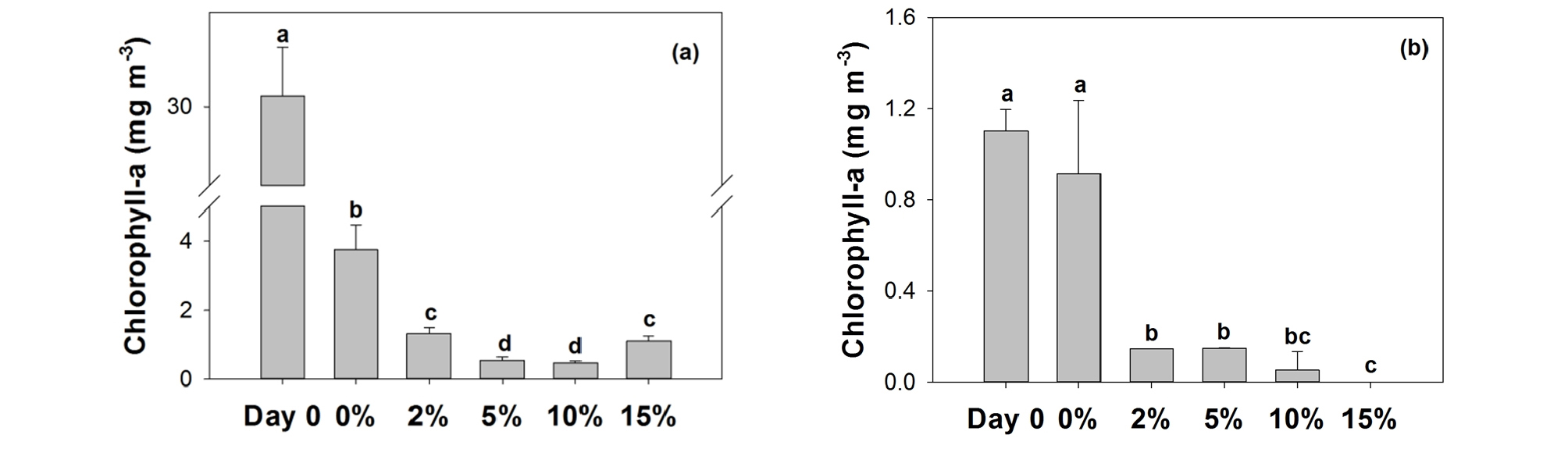
3.1 Chlorophyll-a
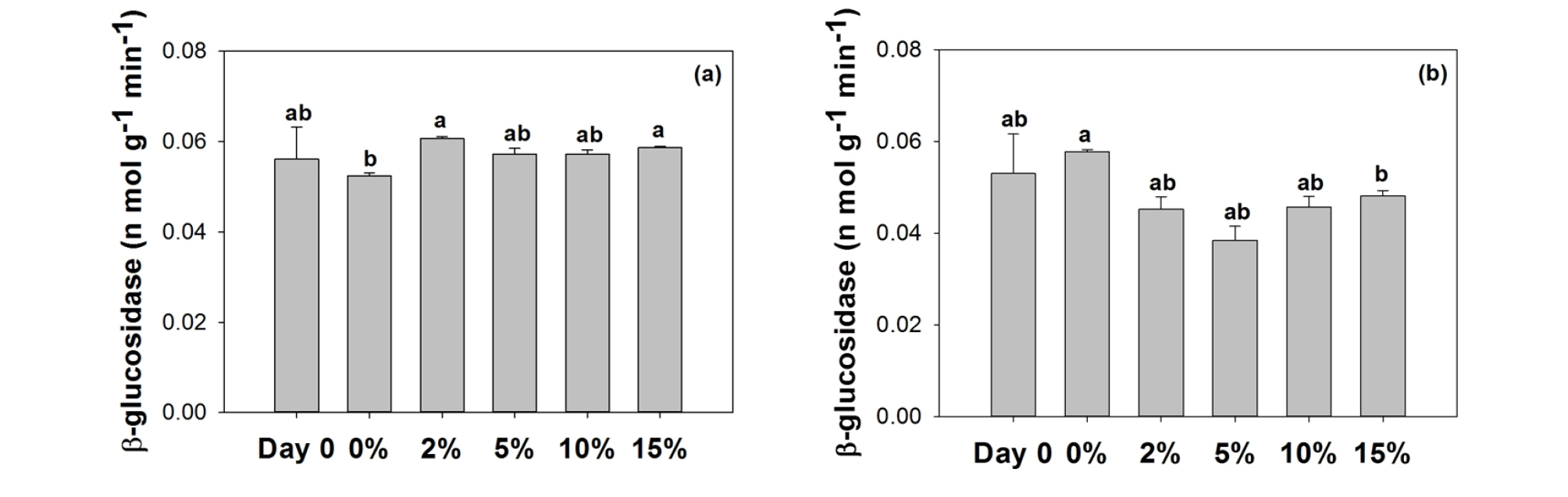
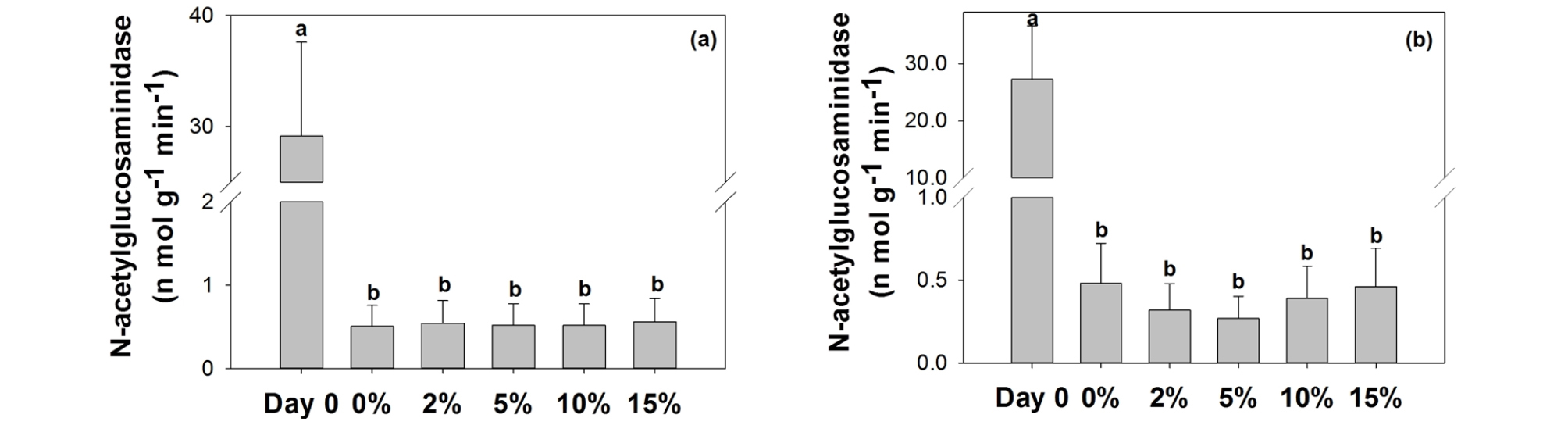
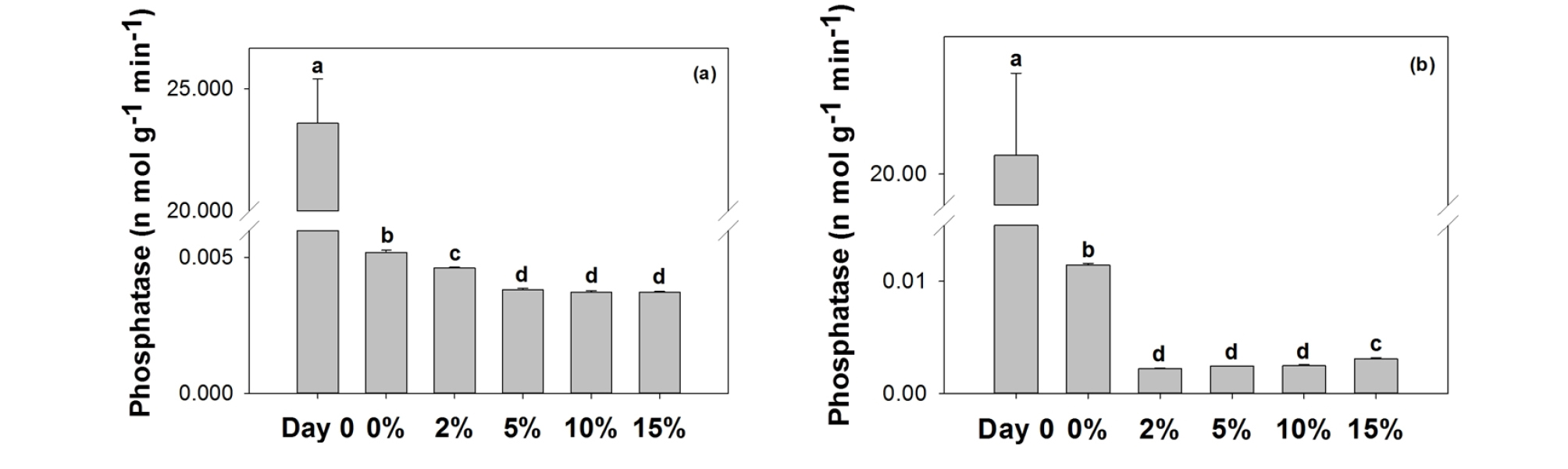
3.2 Extracellular enzymes
3.3 Physical strength
4. Conclusions
1. Introduction
Concrete and mortar have widely used as a building and construction material for their durability and easy applications. However application to water ecosystems such as port, riparian, and lake shores often faces various problems including the growth of attached organisms (e.g., cyanobacteria, algae, moss and shellfish) and organic acid released from them (Warscheid and Krumbein 1996, Sand 1997, Warscheid and Braams 2000). It has been widely reported that organic acids release and physical expansion of growing organism reduce the stability of the surface of concrete materials and hence reduce the durability of overall structure (Ortega-Calvo et al. 1995). Additionally, proliferation of organism on the surface often decreases aesthetic value of structure and water quality in the long-term. Such impacts could be aggravated due to more frequent flooding and water level rise. For example, global climate change models predict more frequent and large scale precipitation in inland ecosystems (Easterling et al. 2000) as well as overall sea level rise (Solomon et al. 2009). As such certain structures will be more frequently exposed to water and vulnerable to growth of attached organism.
To address this problem, various functional concretes and mortars have been suggested or developed. For example, Hiroshi et al. (2001) proposed a painting material containing ferrous sulfate (FeSO4) to inhibit algal propagation on concrete surfaces. Silver nano-particles, quaternary ammonium silane-based biocides, silver copper zeolites, silane siloxane mixtures, and pyridine biocide were also proposed to inhibit the growth of algae (De Muynck et al. 2009). However, these techniques are expensive, difficult to manage, and the duration of the anti-algal effect is relatively short. In addition, they may have less structural stability than normal mortars. Many previous studies visually observed the growth of algae and shellfish on the surface of the concrete structures, but only few studies contain quantitative measurement of algae or changes in water quality by the concrete (Dubosc et al. 2001, Barberousse et al. 2007, Escadeillas et al. 2007). In addition, detailed mechanism for anti-algal function has not been fully understood yet.
Algae and microorganisms on the surface of concrete structures produce extracellular enzymes. Once the enzymes are released from microbes or algae, extracellular enzymes degrade large organic matter into small carbon or inorganic nutrients. They, in turn, are assimilated by microbes and algae to maintain nutrient cycle and energy flow. It has been reported that such enzymatic process is a rate limiting step in overall material cycles. Particularly, phosphatase has drawn much attention in aquatic ecosystems since it releases inorganic phosphorus from organic moiety, which is often a limiting nutrient for algal growth. As such, lowering of phosphatase is needed to secure the long-term inhibition of algal propagation.
In the present study, we employed a new approach using glass beads containing anti-algal property. We prepared mortar specimen containing glass beads with copper compound, which was designed to be released and function as an inhibitory chemical against algae. The specimen was exposed to freshwater or seawater with a large amount of algae, and changes in algal biomass as chlorophyll-a (Chl-a) content and other biological property such as extracellular enzyme activities were monitored.
The goal of this study is to monitor the changes in algal biomass in water that are contacted with mortar with different concentration of copper-containing glass beads and to determine changes in extracellular enzyme activities in water.
2. Materials and Methods
2.1 Mortar composition
Each mortar specimen measures 50 mm × 50 mm × 50 mm, which was prepared by mixing Portland cement (85 g), dried sand (208.33 g), and water (41.225 g) to cure for 2 days. The glass beads were composed of 15Cu2Oㆍ15Na2Oㆍ5B2O3ㆍ65P2O5 of which size ranged between 250-425 µm. This allows rapid dissolution when they are exposed to water. Mortar specimen with five concentrations of glass beads were prepared; 0, 2, 5, 10, and 15%.
2.2 Incubation
Freshwater was collected from Hongje stream in Seoul (37°34’4” N, 126°54’59” E), while seawater sample was collected from Jebudo in Gyeonggi Province (37°10’32” N, 126°37’34” E). Water samples were filtered to remove any suspended materials and l L of water sample was placed in a 2 L container. Mortar specimen with different glass bead concentration was placed in each container and incubated for 37 days. The experiment setting was exposed to 12/12-hour day/night cycle at 20°C to ensure optimal conditions for photosynthesis. Three replicate settings were prepared for each concentration. Water samples were collected at days of 0 (just before the placement of mortar), 5, 10 and 37. Algal biomass and enzyme activities were determined for all concentrations of mortar at day 5, but only control, 2% and 5% settings were determined at all sampling dates.
2.3 Chlorophyll-a
Chl-a concentration was measured by a standard method. In short, 100 mL of water was filtered (GF/C 45 nm) and the filtered paper was ground with 10 mL of acetone solution (9:1). This, then, was maintained at 4°C in dark condition for 24 hours. Extract was centrifuged and absorbance of supernatant was determined at wavelengths of 663 nm, 645 nm, 750 nm, and 630 nm to calculate the concentration of Chl-a.
2.4 Extracellular enzymes
β-glucosidase, N-acetylglucosaminidase, and phosphatase activities were determined using methylumbelliferyl (MUF) compounds as a model substrate. MUF-β-glucopyranoside (400 µM), MUF-acetylglucosamine (400 µM) and MUF-phosphate (800 µM) solutions were used for substrates for β-glucosidase, N-acetylglucosaminidase, and phosphatase, respectively. 5 mL of each substrate solution was added with 1 mL of water sample and incubated for 60 minutes, after which fluorescence was determined by a fluorometer (FLUOstar OPTIMA; BMG Labtech).
2.5 Compressive strength
To determine applicability of mortar prepared with glass beads, compressive strength was determined by a tester (Instrong; Atex).
2.6 Statistical analysis
Basic statistical analyses were conducted by using SPSS 23 (SPSS inc., Chicago, IL, USA). The differences of Chl-a and enzyme activities among treatments were analyzed using one-way ANOVA.
3. Results and Discussion
3.1 Chlorophyll-a
At the end of 5-day’s incubation, concentrations of Chl-a decreased substantially both in freshwater and seawater placed with glass bead mortar (Fig. 1). This result indicates that the mortar proposed in this study did exhibit anti-algal functions. In freshwater system, the presence of mortar itself decreased Chl-a substantially (Fig. 1 (a)), suggesting chemicals in mortar itself is highly toxic to freshwater algae. It is speculated that freshwater algae are highly sensitive to pH changes by mortar. In contrast, such decrease was not observed in seawater (Fig. 1 (b)), probably due to higher resistance of seawater algae to higher pH as they are exposed to alkaline pH in natural conditions.
Copper is known to inhibit both seawater and freshwater algae at as low concentration as 1 ppb (Nielsen et al. 1971). Copper can be incorporated into algal cell to influence physiology as well as inhibition of sulfide residues of key enzymes. Additionally, it has been reported that copper can interfere with biosynthesis of pigments and phospholipids, resulting in lower photosynthetic rates (Barón et al. 1995).
3.2 Extracellular enzymes
No distinctive changes were found in ß-glucosidase activities by glass bead containing mortar in freshwater (Fig. 2 (a)), while the activities decreased slightly by glass bead containing mortar in seawater (Fig. 2 (b)). However, no differences were discernible in relation to the concentrations of glass beads. β-glucosidase is involved in organic carbon (i.e., cellulose) decomposition, and this result suggests that impacts on carbon cycling would be minimal by the mortar. Another possibility is that algal biomass would die off and provide ample amount of organic carbon to systems, which may mask-off inhibitory role of glass beads on β-glucosidase activity.
Similarly, N-acetylglucosaminidase activities did not change by the presence of glass beads (Fig. 3). This enzyme is involved in chitin decomposition and hence reflects influences on nitrogen and partially carbon cycles. The results suggest that nitrogen cycle would not be strongly affected by glass bead in the systems. It is interesting to note that mere presence of mortar decreased dramatically N-acetylglucosaminidase, indicating that certain components in mortar or environmental changes by it could impede nitrogen cycle in the long-term. Further studies are warranted to reveal mechanism for such intense inhibition.
Unlike β-glucosidase or N-acetylglucosamindase activities, mortar with glass-beads did decrease phosphatase activities in both systems (Fig. 4). A stronger inhibition was found in seawater system compared with a freshwater ecosystem. This result is in accordance with Wang et al. (1995), who reported inhibition of alkaline phosphase by copper in marine algae. This result has an implication that glass bead with copper would not only inhibit directly algal propagation (as expressed in Chl-a content), but also may hinder its proliferation by impeding phosphorus cycle. Since P is often the most common limiting factor for algal production in aquatic ecosystems, decreases in phosphatase would induce slower P release and lower algal production.
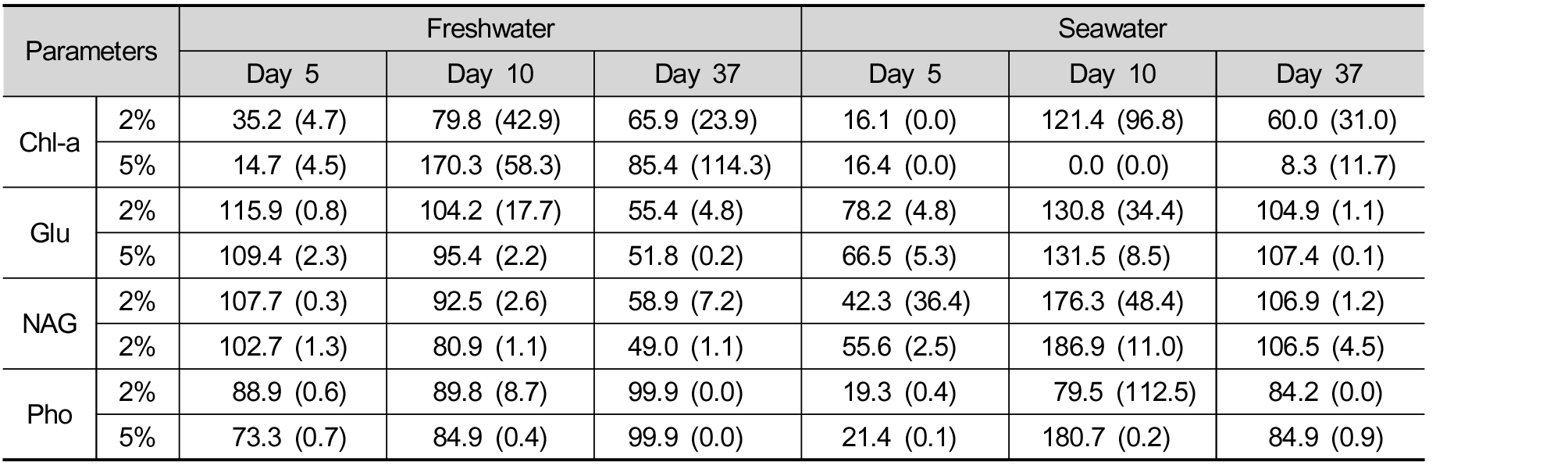
If data for 37 days’ incubation were all considered, some parameters exhibit different patterns from those at 5 day. For example, substantial decreases in Chl-a at day 5 disappeared in freshwater ecosystem, while such inhibition lasted in seawater system (Table 1). Particularly, 5% mortar in seawater maintained strong inhibitory effects on algal biomass over 37 days. Similarly, lowered phosphatase activities by glass beads recovered to the level of control in freshwater system (Table 1). In contrast, β-glucosidase and N-acetylglucosaminidase activities decreased in the longer-term compared with those at day 5. A mortar proposed in this study would maintain function properly in seawater system, while long-term consequence may differ in freshwater system.
3.3 Physical strength
Compressive strength of mortar specimen ranged between 27.64-28.91 MPa, and no significant differences were found among different treatments. This result indicates that functional mortar proposed in this study has the same physical strength as control mortar without glass beads.
4. Conclusions
Functional mortar with Cu glass exhibited lower Chl-a content, suggesting that the functional mortar reduced algal growth, while phosphatase activity also decreased. Concentration of DOC increased because debris of dead algae increased. The overall results suggest that mortar with copper glass beads would exhibit anti-algal property. Particularly, it would be more effective for seawater than freshwater systems. In addition to direct effects on algal proliferation, copper has a potential as an inhibitory material against phosphatase, which plays a key role in P cycle and algal production. Further study should focus on the longer-term consequence of functional mortar proposed in this study, probably by exposing them under field conditions.