1. Introduction
Among traditional water treatment processes, heterogeneous photocatalytic oxidation processes using semiconductor particles such as titanium dioxide (TiO2)-based materials and other oxides have been found to be a promising solution to remediate water resources (Chong et al. 2010, Di Paola et al. 2012, Doria et al. 2013, Joo et al. 2013, Jang et al. 2016). While many TiO2-based photocatalysts have been developed with bare TiO2 or modified TiO2 particles, suspended and fine photocatalyst particles in a slurry system for water treatment would require several processes (i.e., separation, recycling, and regeneration) in large-scale water treatment processes. To avoid the particle recovery processes, photocatalyst particles can also be immobilized and stabilized onto various supports (i.e., silica gel, glass fibers and beads, activated carbons, mesoporous clays, cellulose membranes, polymer films, etc.) (Mozia 2010, Doria et al. 2013, Joo et al. 2013, Leong et al. 2014, Jang et al. 2016). However, immobilizing photocatalysts to various supports reduces the amount of active sites, enlarges the mass transfer limitations, and increases the operation difficulty with reduced photon penetration (Chong et al. 2010, Di Paola et al. 2012).
Considering these limitations and challenges when developing efficient photocatalyst/support composites, the physicochemical bonds between certain photocatalysts and supports must be strong without reducing the photoactivity. Recently, expanded polystyrene (EPS) has been used in shock absorbing packing materials, and has also several other advantages such as good flexibility, greater resistance to mechanical fatigue and chemicals, light density, low melting point (i.e., 120°C) and non-toxicity (Ross and Evans 2003, Lee et al. 2010). Although various coating and immobilization techniques (i.e., sol-gel, chemical vapor deposition, reactive magnetron sputtering, spray pyrolysis etc.) have been already developed (Lee et al. 2010), these coating and immobilization techniques are expensive and impractical. Therefore, in this study, a simple immobilization technique using nanoscale TiO2 powder to EPS balls with temperature- controlled melting method was developed, and the photocatalytic activity of TiO2 powder-embedded EPS balls was evaluated using methylene blue (MB) solution under ultraviolet irradiation (principal λ = 254 nm). The specific objectives of this study were (1) to develop a new immobilization technique of nanoscale TiO2 powder to expanded polystyrene (EPS) balls with a temperature-controlled melting method, and (2) to elucidate the photocatalytic activity of TiO2 powder-embedded EPS balls using a methylene blue (MB) solution under ultraviolet irradiation (λ = 254 nm).
2. Materials and Methods
Commercial low-density uniform EPS balls for the supports were purchased (HUG Co., Republic of Korea). The diameter of low-density uniform EPS balls ranges around 1-10 cm, and the diameter of 1 cm balls was selected for lab-scale batch photocatalytic reactor (BPR) used in this study. As a representative photocatalyst, P25 TiO2 (Evonik Degussa GmbH, Germany) was purchased from Skybright group (Hong Kong). Glycerin was used as the dispersing medium of TiO2, and was purchased (Daejung Chemicals & Metals, Republic of Korea). A suspension solution of TiO2 was prepared by dispersing 1,000 g of TiO2 in 5 L of glycerin over 70°C using a heating pan with a digital thermo-regulator and a magnetic stirrer.
After complete dispersion of the TiO2 in 5 L of glycerin solution, the suspension solution was heated up to the melting point of the EPS balls at around 140-145°C. Then, the EPS balls were repeatedly soaked in suspension solution of TiO2 for 5 seconds to immobilize the TiO2 particles over the surface of the EPS balls. After repeated the soaking processes several times, the EPS balls were covered with a thin film of TiO2 suspension. Then, EPS balls were covered with clear commercial wrapping vinyl. Clear vinyl-wrapped EPS balls were completely soaked in cold water (1-4°C) to enhance the attachment of TiO2 particles on the surface of the EPS balls. Finally, TiO2-immobilized EPS balls were rinsed with distilled water using an ultrasonic cleaner (POWER SONIC 420) (Hwashin Tech, Korea) at 700 W for 60 min to remove any excess TiO2 particles not trapped on the surface of the EPS balls. After rinsing, TiO2-immobilized EPS balls were air-dried at room temperature. The whole production procedures of the TiO2-immobilized EPS balls used in this study were displayed in Fig. 1.
The surface of both intact and TiO2-immobilized EPS balls was characterized by scanning electron microscopy (SEM) and energy dispersive X-ray spectroscopy (EDS) (JEOL, Germany). The photocatalytic activities of TiO2-immobilized EPS balls were analyzed based on the degradation efficiencies of MB in a BPR consisted of a rectangular mirror-coated PVC plastic and 4 UV-C lamps (8 W, 254 nm in wavelength) as shown in Fig. 2. The initial MB concentration of 10 mg/L was prepared by dissolving 5 mg of MB into 500 mL of distilled water. Degradation experiments of MB were performed using four quartz reactors as follows: 1. UV only (photolysis), 2. TiO2-immobilized EPS balls with UV (photolysis & photocatalysis), 3. intact EPS balls with UV (photolysis & sorption), 4. TiO2-immobilized EPS balls without UV (sorption). Every 10 minutes for an hour, a 1.0 mL of aqueous sample was collected. To evaluate the life span of TiO2-immobilized EPS balls, used TiO2-immobilized EPS balls were washed and air-dried to repeat the same degradation experiments several times.
3. Results and Discussion
3.1 Characterization of TiO2-immobilized EPS balls
The scamming electron microscope (SEM) images of the newly-developed TiO2-immobilized EPS balls at two different magnifications (i.e., x 50 and x 500) and SEM/EDS analysis are shown in Fig. 3. As shown in Fig. 3, the surface of intact EPS balls was relatively smooth and non-porous, whereas the surface of TiO2-immobilized EPS was relatively rough and porous with relatively homogeneous patches of TiO2 and glycerin film on the surface. Based on the associated EDS analysis, the components of intact EPS balls are carbon and oxygen whereas those of TiO2-immobilized EPS balls are carbon, oxygen, and titanium, indicating that relatively homogeneous patches of TiO2 were immobilized on the surface of EPS balls.
3.2 Degradation of methylene blue using TiO2-immobilized EPS balls
After batch experiments of MB were performed using UV only (photolysis), intact EPS balls with UV (photolysis & sorption), TiO2-immobilized EPS balls with UV (photolysis & photocatalysis & sorption), TiO2-immobilized EPS balls without UV (sorption) in BPR, the removal efficiencies of MB in the aqueous phase were evaluated. As shown in Fig. 4, the degradation of MB using only UV was insignificant, indicating that photolysis of MB in the aqueous phase was found to be negligible. Although the sorption of MB to both intact and TiO2-immobilized EPS balls without UV was slightly observed, the sorption amounts and intensities of MB were insignificant due to the thermodynamically unfavorable sorption processes. Considering that no interactive reactions between MB molecules and EPS balls with low porosities and low surface area occurred, the mass transfer of MB in the aqueous phase to non-crystalline polymer is insignificant.
When comparing the MB removal efficiencies between photolysis (UV only) and photocatalysis (UV + TiO2-immobilized EPS balls), the removal efficiencies were significantly improved with the TiO2-immobilized EPS balls under UV irradiation. Because heterogeneous photocatalytic degradation of MB occurred on the surface of TiO2-immobilized EPS balls, highly reactive transitory species effectively degraded ambiguous refractory MB molecules, indicating that the surface reactions in heterogeneous photocatalysis were more dominant than those of photo-induced photolysis in aqueous solutions. Thus, TiO2-immobilized EPS balls were found to be an effective photocatalyst for photodegradation of MB in aqueous solutions. Therefore, buoyant TiO2-immobilized EPS balls developed in this study can be used to treat contaminated surface water on site although further studies are warranted to evaluate the durability of photoactive TiO2 on the surface of EPS balls.
3.3 Effect of sonication on the degradation of methylene blue using TiO2-immobilized EPS balls
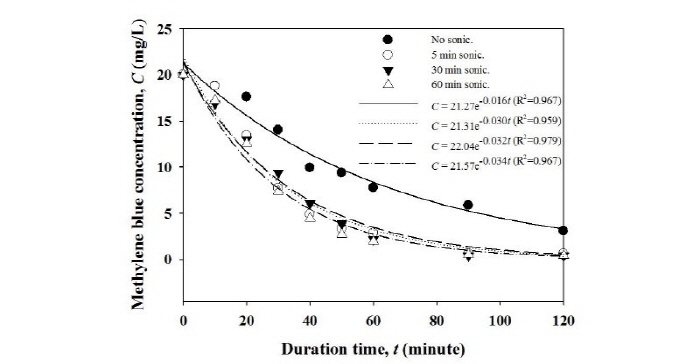
When comparing the MB removal efficiencies before and after sonication, the removal efficiencies were significantly improved after sonication (see Fig. 5). These results could be the breakage of the glycerin film coated on the surface of EPS balls caused by sonication, which makes more TiO2 particles exposed to the MB molecules and UV irradiation. After sonication, more TiO2 particles on the EPS balls can be involved into the photocatalytic reactions, and can increase the MB removal efficiencies. As is also evident by Fig. 5, the degradation constant (k) increased from 0.016 to 0.034 min-1, indicating that sonication can enhance the photocatalytic reactions of TiO2 particles on the surface of EPS balls. However, the time of sonication is not a significant factor to influence the degradation of MB. Thus, further study is in progress to elucidate whether the breakage of the glycerin film increases the exposed surface area of TiO2 particles immobilized on the surface of EPS balls.
4. Conclusions
In this study, a new immobilization technique of nanoscale TiO2 powder to expanded polystyrene (EPS) balls with temperature-controlled melting method was developed. Under ultraviolet irradiation (λ = 254 nm), the photocatalytic activities of TiO2 powder-immobilized EPS balls were evaluated using a methylene blue (MB) solution. Based on the surface characterization analysis results, the components of intact EPS balls are mainly carbon and oxygen, whereas those of TiO2-immobilized EPS balls are carbon, oxygen, and titanium. Also, relatively homogeneous patches of TiO2 and glycerin film were coated on the surface of EPS balls.
Based on a comparison of degradation efficiencies of MB between intact and TiO2-immobilized EPS balls under UVC illumination, the degradation efficiencies of MB were significantly improved using TiO2-immobilized EPS balls, and surface reactions in heterogeneous photocatalysis were dominant relative to photo-induced radical reactions in aqueous solutions. Thus, TiO2-immobilized EPS balls were found to be an effective photocatalyst for photodegradation of organic compounds in aqueous solutions without further processes (i.e., separation, recycling, and regeneration of TiO2 powder).