1. Introduction
2. Materials and Methods
2.1 Experimental design and set-up
2.2 Characterization of soil sample and treatment of P and Fe
2.3 Chemical assay with single extraction method
2.4 Biological assay with lettuce
2.5 Statistical analysis
3. Results and Discussion
3.1 Soil properties
3.2 Effects of Fe and P on As extractability
3.3 Effects of Fe and P on root elongation in lettuce
3.4 Correlation analysis for lettuce growth and its As and P uptake
3.5 Confirmation study
4. Conclusion
1. Introduction
Tailings containing high levels of arsenic (As) that originated from mining activities have been recognized as a principal anthropogenic source of As contamination of soil (Bruce et al. 2003; Seidel et al. 2005). Many other studies related to the remediation of As-contam-inated soil have reported that chemical stabilization using various types of amendments has been identified as a lower cost and lower input method than other physical and chemical methods (Lee et al. 2009; Koo et al. 2013). Kumpiene et al. (2008) also reported that the type of amendments and those of mechanisms of As stabilization. Among the amendments for As stabilization in soil, iron (Fe) sources have been known to be effective and common agents and have been more widely used than other amendments (Bowell 1994; Warren and Alloway 2003; Lee et al. 2011; Koo et al. 2012).
For real remediation, not only should As be stabilized but re-vegetation should also be accomplished. Vege-tation could promote the coverage of the tailing area, resulting in a decrease in soil erosion and As hazards to humans and an improved landscape and pollution management (Tordoff et al. 2000; Conesa et al. 2007). Soils contaminated with mine tailings are not suitable for plant growth because of macronutrient deficiencies (Tordoff et al. 2000; Koo et al. 2013). Hence, many agents, such as pH control agents, carbon sources, and phosphorus (P) have been applied to enhance soil fertility (Chen et al. 1998; Heeraman et al. 2001; Juwarkar et al. 2008).
Because As and P have similar chemical charac-teristics in soil, competition for adsorption sites occurs (e.g., Fe - (hydro) oxide and organic matters); thus, the mobility of As in soil has been increased by the input of P-rich agents (Woolson et al. 1973; Cao et al. 2003). Cao et al. (2003) reported that P amendments signifi-cantly increased soil water-soluble As, resulting in the enhancement of As uptake by plants. Furthermore, Cao and Ma (2004) found that the incorporation of phosphate in soil increased As accumulation in carrots and lettuce resulting from an increase in soil water- soluble As because of the replacement of As by phosphate in soil. Additionally, Davenport and Peryea (1991), via a column experiment, found that the addition of P fertilizer significantly increased As leaching from soil. To identify specific interactions among As, P, and Fe, Koo et al. (2013) demonstrated the effects of P and Fe in As-spiked soils via a sequential incorporation method for Fe and P using response surface methodology. They found that the P content of soil affected the reduction of As toxicity towards the root elongation of lettuce by providing a nutrient source rather than by suppressing the uptake of As. The soil Fe content also caused root elongation by reducing the mobility of As. Nevertheless, adding P and Fe to As-contaminated soils stimulated plant growth during the early stage of root growth; the experiment was limited to the use of artificial soil. Unlike artificial soil, various factors in As-contaminated field soil affect As mobility (e.g., the pH value, redox potential, metal oxides, organic matter, and clay minerals) but the related studies are insufficient (Bissen and Frimmel 2003; Signes-Pastor et al. 2007; Hossain et al. 2008).
Thus, the purpose of this study was to investigate the effects of P and Fe on the availability of As and the root elongation of lettuce (Lactuca sativa L.) in As-contaminated soil using response surface meth-odology. The model was used for the interpretation of the effects of P and Fe on As mobility and As accumulation in lettuce.
2. Materials and Methods
2.1 Experimental design and set-up
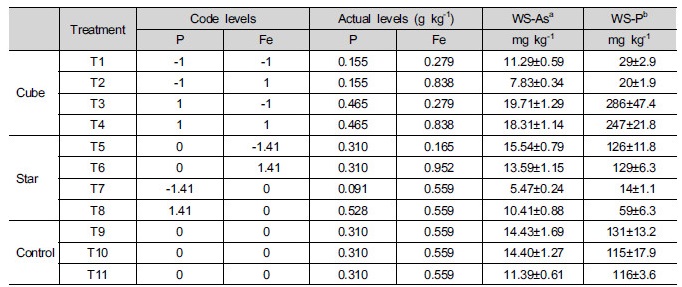
In the present study, the second-order central com-posite rotate design (CCRD) was adopted and the values of parameters selected for CCRD are summarized in Table 1. The studied model (3 experimental points and 11 experiments) required for two factors and five levels in the CCRD, which were determined as follows: 2n (22 = 4: cube points) + 2n (2 × 2: star point) + 3 (central points: three replicates) (Son et al. 2009, Koo et al. 2011). Low and high factors (cube points) are coded as -1 and 1, the mid factor settings (central points) coded as 0, and the center points of the tube (star points) are coded as -1.41 and 1.41 (Hao et al. 2006).
Using multiple regression analysis of the experimental results, the following response Y was calculated through the second-order polynomial model:
Y = β0 + β1χ1 + β2χ2 + β12 χ1χ2 + β11 χ12 + β22 χ22
(Eq. 1)
where Y contains the dependent variables (As mobility and bioavailability using chemical extraction methods, the root elongation of lettuce, and the amount of As and P in lettuce root and shoot), β0 is the intercept, βi the linear coefficient, βij the interaction coefficient, βjj the quadratic coefficient, and χ1 and χ2 (for P and Fe contents, respectively) the coded independent variables. The experimental design of the present study de-veloped mathematical models for explanation of the results. The generated model also was used in simulations for providing useful information on the effects P and Fe on As mobility and toxicity towards lettuce growth.
2.2 Characterization of soil sample and treatment of P and Fe
Surface soil contaminated with mine tailing was taken from the Gangwon mine, a former gold mine site at Gangwon province, the Republic of Korea (37°19'19.6", 128°48'47.4") at November, 2014. The soil sample was air dried and passed through a 2 mm sieve. The soil pH and electrical conductivity (EC) were determined using 1:5 soil:water suspension with a combination pH-EC meter (Thermo Orion 920A, USA). The total phosphorus was determined followed by HClO4 digestion methods and the concentration of P in filtrates was determined using an ultraviolet (UV) spectrophotometer (UV-1650PC, Shimadzu, Japan), via a modified molybdenum blue method, which removed the arsenate interference by reducing the arsenate (V) to arsenite (III) using dithionite (S2O42-) (Tsang et al. 2007). The available phosphorus in soil was measured using the Mehlich-3 solution (Mehlich 1984). The concentration of P in filtrate was also determined with the same previous method. The total As concentration was determined by digesting samples with aqua regia, a mixture of HNO3/HCl (v:v = 1:3), using an inductively coupled plasma-optical emission spectrometer (ICP-OES) (Vista Pro, Varian USA). The Fe, Al, and Mn concentrations were determined followed by ammonium oxalate extraction methods (Loeppert and Inskeep 1996).
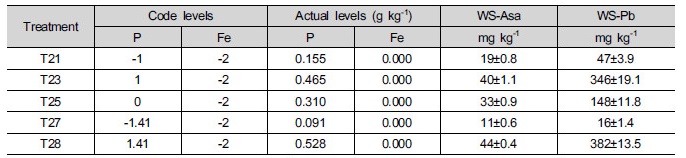
Eleven different soil compositions were provided by adjusting the P and Fe contents to levels determined by the CCRD (Table 1) considering the results from former study (Koo et al. 2013). P (ranging from 0.091 to 0.465 g kg-1) and Fe (ranging from 0.165 to 0.952 g kg-1) were added in the forms of Na2HPO4 (Sigma- Aldrich, USA) and zero-valent iron (ZVI, obtained from Sigma-Aldrich), respectively. After treatment, the soils were equilibrated at room temperature and 60% of the water holding capacity (WHC) for 4 weeks prior to the chemical extraction and phytotoxicity tests. In addition, five confirmation experiments were subsequently performed to support the findings resulting from the CCRD study (Table 2).
2.3 Chemical assay with single extraction method
The effect of P and Fe on As extractability was evaluated using deionized water (DW). For water soluble-As, 1 g soil was added to 20 mL DW in 50 mL polypropylene centrifuge tubes (Falcon, London, UK), which were then shaken for 1 hour on a wrist- action shaker, centrifuged for 20 min at 5100 rpm, and then filtered with a PTFE 0.45 um pore size syringe filter (Rodriquez et al. 2003). All the remaining As and P contents in the filtrates were determined using ICP-OES after acidification (Sikora et al. 2005).
2.4 Biological assay with lettuce
The root elongation tests using lettuce (Lactuca sativa L.) were conducted according to the OECD Guidelines for the Testing of Chemicals (OECD 208 2006). Prior to sowing, the seeds were sterilized with 10% H2O2 and subsequently 12 seeds were placed in 100 × 20 mm plastic Petri-dishes which containing 40 g of the treated soils. The dishes were placed in a growth chamber randomly. The moisture content was controlled at approximately 60% of the WHC of the soils, with the light conditions of 16 hours of daylight and 8 hours of darkness per day, at 20 ± 2°C. Four weeks after sowing, the plants were harvested to determine their root growth using an image analyzer program (WinRhizo 5.0a, Regent, Canada). The root elongation was calculated for each Petri-dish based on the each root length of plant and expressed as mm seedling-1. After root elongation determining, the plants were separated into roots, shoots and dried in an oven at 70°C. The dried samples were digested with HNO3 and H2O2 by hot-block digestion procedure, and immediately filtered. The filtrates were used to determine the concentrations of As using ICE-OES. The concentration of P in filtrates was determined using same method for total phosphorus analysis (section 2.2).
2.5 Statistical analysis
The statistical analysis of the results was performed using the SAS program (SAS 9.2, USA). The rela-tionships among the results were evaluated using a Pearson’s correlation analysis at p<0.05. The parameters and their significances in the second-order polynomial models were tested using the multiple regression procedure and fitted to a second-order polynomial model.
3. Results and Discussion
3.1 Soil properties
An As analysis indicated that the soil was highly contaminated by As (1,854 mg kg-1), resulting in the exceedance of the ‘regulatory level’ for As (75 mg kg-1) described in the Soil Environmental Conservation Act of South Korea (KMoE 2011). The pH and EC of soil were 8.41 and 0.76 ds m-1, respectively, a slightly basic pH. The total and available phosphorus (P) concentrations of 1.10 and 0.07 g kg-1, respectively indicated that the available form of P was low (6.36%), so the effect of available P on As mobility in the soil was also low. The oxalate extractable Al (Alox), Fe (Feox), and Mn (Mnox) concentrations were 1.18, 13.4, and 2.0 mg g-1, respectively. The highest Feox was observed in a soil sample, indicating that the main adsorption sites for As is in soil.
3.2 Effects of Fe and P on As extractability
The application of P and Fe had an effect on the As extractability in soil and water soluble-As and P (Table 1). A model fitting the results from Table 1 was constructed with a regression analysis. The p value in analysis of variance (ANOVA) was less than 5% of the estimated F value (7.2) obtained for the water soluble-As extractability, indicating that the model was significant at a high confidence level (95%). The correlation coefficient, r2, was 0.474, and the model applied to the data yielded the following equation:
The water soluble As (mg kg-1) = 13.10 + 3.24 × P – 0.95 × Fe (Eq. 2)
The model provided significant information for deciding which soil parameter was important for As mobility. As expected, a significant and positive coefficient was obtained for the P content and the Fe content had different results than that of P. In addition, the results showed that the effect of P on the changes in As mobility was greater than that of Fe by comparing the estimated coefficients for P (3.24) and Fe (0.95) (Eq. 2). According Cao and Ma (2004), the application of phosphate in soil increased the water-soluble As because of the replacement of As by phosphate in the soil. Additionally, Fe is well known to reduce As mobility in soil via the adsorption of As onto its surface through previous studies (Bowell 1994; Warren and Alloway 2003; Lee et al. 2011; Koo et al. 2012). In this regard, the changes in As mobility were expected to directly affect plant As uptake and toxicity.
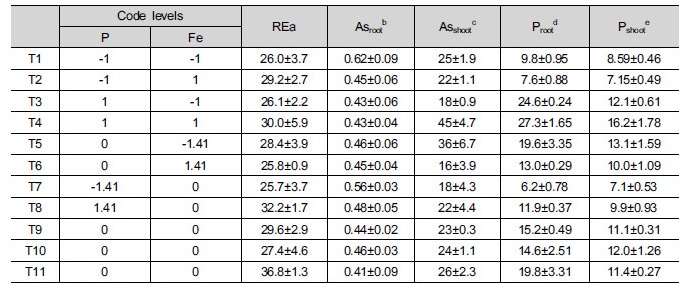
3.3 Effects of Fe and P on root elongation in lettuce
The simultaneous application of P and Fe also affected root elongation in lettuce (Table 3). The p value in ANOVA for the root elongation of lettuce was not less than 5% of the estimated F value (3.80) , indicating that the result was not significant, and the calculated correlation coefficient, r2 = 0.3221, was poor. Although the data did not show significant results, the developed model yielded the following equation:
The root elongation of lettuce (mm seedling-1) = 28.40 + 2.18 × P (Eq. 3)
There was no doubt that Fe affected both the As mobility and lettuce root growth. Nevertheless, the model provided information that only the addition of P affected lettuce root elongation even though both P and Fe were added. According to a similar study by Koo et al. (2013), both P and Fe affected lettuce root growth significantly, and the estimated coefficient for P (185.81) was higher than that for Fe (92.91). This result might be explained because the effectiveness of P was much greater than that of Fe. Moreover, the present study used As-contaminated soil unlike the previous study (Koo et al. 2013), so other factors (e.g., macro/micro nutrients, dissolved organic carbon, or clay content, other metal hydroxide/oxides) could have affected As mobility and phytoavailability (Cai et al. 2002; Fitz and Wenzel 2002; Koo et al. 2011). Additionally, this result contradicted our earlier as-sumption that the addition of P increases As mobility, which causes an increase in phytotoxicity. On the contrary, the addition of P seemed to promote root growth by acting as a nutrient rather than a toxin to increase As mobility. From these results, it was concluded that in As-contaminated soil, the application of P increases soil nutrient levels, promoting lettuce root growth. To interpret and demonstrate the results, more data will be discussed below.
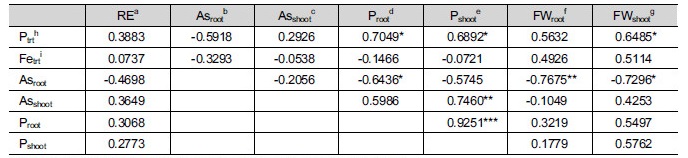
3.4 Correlation analysis for lettuce growth and its As and P uptake
The relationships among the results were evaluated using Pearson’s correlation analysis (Table 4). As expected, a negative coefficient (r = 0.4698) was found only between the effect on the lettuce root elongation and the As content in lettuce roots. It is known that the toxic effects of the absorbed As on root growth are caused by oxidative stress induced by an increase in the production of reactive oxygen species (ROS). ROS might cause the inhibition of root elongation by disrupting cellular activity or the interference with plant cell metabolism (Gratão et al. 2005; Koo et al. 2011). The correlation coefficients between root elongation and other indices indicated that the As content in the roots seemed to be the main reason that root growth was impeded, even though the result was not significant. This statistical result resulted from the complexity and variety of the examined field soil compared to that in the previous study using artificial soil (Koo et al. 2013).
The absorbed As content in lettuce roots was decreased by increasing the amount of P (r = -0.5918) and Fe (r = -0.3293) (Table 4). In general, Fe (hydro) oxides have a high affinity for the adsorption of As; hence, the As mobility in soils is reduced with the presence of Fe on the soil surface (Codling and Dao 2007; Koo et al. 2013). Rahman et al. (2013) reported that Fe2+ input decreased the As concentration in the roots of rice seedlings in paddy soil despite the uptake of As by the rice roots via a phosphate uptake pathway due to the similar physicochemical characteristics of Fe2+ and As (Liu et al. 2004). In addition, the use of Fe0 in As contaminated soil also reduced the accumulation of As in Panax notoginseng (Burk.) roots (Yan et al. 2013). The As concentration in radish roots was significantly decreased by approximately 50% by the addition of steel-making slag (SMS) compared to the control (Qutierrez et al. 2010). For P, as might not be expected, the As concentration in lettuce roots also decreased with an increase in P. This result agreed with a previous study (Koo et al. 2013) that showed that the As content in lettuce roots was negatively related to the P concentration in artificial soil (r = -0.459). These similar results were obtained because of competition between As and P for the same P transporters in plasma membrane of root cells (Ullrich- Eberius et al. 1989; Meharg and Hartly-Whitaker 2002; Gunes et al. 2009). This interpretation was also applicable to another correlation between the As and P concentration in lettuce roots (p = -0.6434). Because As and P competed when uptake occurred near the root surface, both As and P interfered with each other.
The P concentration in roots and shoots increased significantly (p = 0.7049 and p = 0.6892, respectively), and a high correlation coefficient was found between roots and shoots (p = 0.9251) (Table 4). P is a plant growth macronutrient and is involved in key functions, such as structural cell components (e.g., phospholipids and nucleic acids), and in plant metabolism (e.g., energy transfer, photosynthesis, nutrient movement within plants) (Cao et al. 2003; Knudson et al. 2003; Rufyikiri et al. 2006). Thus, the fresh weights of roots and shoots were positively related to the P treatment concentration (r = 0.5632 and r = 0.6485, respectively). Conversely, the absorbed As content in roots decreased the fresh weight of roots and shoots, significantly (r = -0.7675 and r = -0.7296, respectively), indicating that the uptake of As by roots might suppress root elongation, leading to a decrease in the weight of the lettuce.
3.5 Confirmation study
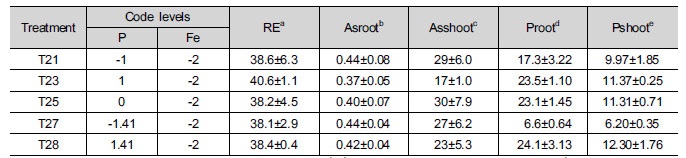
Additional experiments (Table 5) were conducted to verify the effectiveness of Fe followed by P treatment. The treatment concentration of P in T21, T23, T25, T27, and T28 were same as T1, T3, T5, T7 and T8, respectively, without any Fe treatment. Therefore, the only effects of P in the absence of Fe occurred for As and P extractability and As and P absorption into lettuce. Compared to table 1 (T1, T3, T5, T7, and T8), table 2 (T21, T23, T25, T27, T28) showed that the concentration of water soluble As and P increased in the absence of Fe and that the degree of increase was higher for P than for As, indicating that the available P was increased. The changes in the extractability increased the absorption of P, but decreased that of As by lettuce roots, leading to an increase in root elongation in lettuce (Table 5) compared to Table 3. It seems that the absorption of As by lettuce roots was decreased due to the suppression of P in plasma membrane of root cells, which was same as earlier results. Conversely, these results indicated that although the addition of Fe decreased the extractability of both As and P, the phytoavailability and phytotoxicity were greatly affected by the relative available amounts of As and P in soil.
4. Conclusion
This study evaluated the effects of P and Fe on the extractability of As and P and phytotoxicity in soil highly contaminated by As. Under an incorporation of both P and Fe, the P treatment increased root elongation of lettuce by supplying nutrient source and decreased As absorption in lettuce root by suppressing the uptake of As. Under only the presence of P condition, the P treatment content increased the contents of available P more than that of available As, which resulted in the deceases in As toxicity toward lettuce, more effectively. Nevertheless, only the application of P could cause increases in As mobility, leading deterioration of environmental health. Also this study used limited environmental soil sample and chemical reagents. Thus, further study seems to be needed for the application of P and Fe to field scale using various adaptable P fertilizers and Fe sources.