1. Introduction
2. Materials and Methods
3. Results and Analysis
3.1 Effect of Time on Available P
3.2 Effect of Fertilizers on Available P
4. Discussion
1. Introduction
Phosphorus (P) is a macronutrient that is one of the essential nutrients for the proper growth of plants. It comes second to nitrogen as the most limiting nutrient to plant growth. The importance of phosphorus to plants as a nutritional element is wide. P is essential to cell growth and development of tissues, energy and respiration, hardiness and maturity (Baker and David 2015). Human beings and animals require sufficient amount of phosphorus for the body to grow and develop.
Cause of P limitation is mainly the inadequate supply of the element and the losses within cycle. Unlike the nitrogen cycle, phosphorus does not exist within the atmosphere rather it exists as solids in phosphorus compounds either in soil or in rocks. Phosphorus hardly exists in its elemental form because it boasts of a highly reactive nature forming compounds instead, particularly with oxygen, existing as phosphates (Hyland et al. 2005).
To sustain agriculture, fertilizer or other forms of soil amendments that can retain, add or increase phosphorus in the soil are applied. Studies show that the different types of phosphorus amendments added to soil depend on factors such as their comparative efficiencies and factors that affect their need such as soil reaction and nature of crop grown (Delgado et al. 2002).
Based on those factors, farmers mostly deal with three main sources for phosphate amendment; Inorganic fertilizers such as superphosphates, animal based amendments such as manure and by products e.g. meat and bone meal (Delin 2015). In Korea as well as most developed countries, inorganic phosphorus fertilizers are preferred for their fast acting results.
Meat and bone meal/bone meal (MBM/BM) fertilizers boast of appreciable phosphorus amount as same as nitrogen and potassium proving to be useful as fertilizer for many crops (Nogalska 2013). The limitation with MBM is that studies have indicated that it is more effective as a P fertilizer in acidic soils as opposed to those with a pH higher than 6 (Jeng et al. 2007). Addition of fertilizers to a soil changes the composition of the soil solution, which in turn affects the dynamics of soil phosphorus (Delgado et al. 2002).
After the initial addition of a fertilizer, its reaction with the soil water renders it available owing to the solubility and plants easily take it up. However, the solubility decreases with subsequent days as the phosphorus in the fertilizers starts to react with soil elements. Through either adsorption or formation of insoluble compounds such as those formed with aluminium or calcium. The efficacy and efficiency of phosphorus fertilizers is decreased (Pierzynski et al. 2005). The changes lead to a constant need to keep fertilizing the soil every time to add to the labile pool. In turn, each application also adds on to the active. The main disadvantage of phosphorus amendment is that exceed amount of phosphorus has to be disposed of through leaching.
Researchers have studied various methods to boost P fertility using either inorganic, organic or raw rock phosphate in order to retain or recover soil phosphorus. In recent times, researchers have been studying biochar as a possible amendment to use with these fertilizers to evaluate its effect on increasing P availability (Blackwell et al. 2009).
Biochar is defined as the carbon rich product pro-duced in limited amount of oxygen at temperatures less than 700 from organic biomass such as wood or animal waste (Lehmann et al. 2009). Recent studies (Chintala et al. 2014; Chan et al. 2008; Lehmann et al. 2003), have shown the ability of biochar in improving soil quality, having a long turnover time in the soil and increasing crop production. The high surface area and porosity of biochar enables it to adsorb or retain nutrients and water (Chintala et al. 2014), provide habitat for beneficial microorganisms to flourish and reduce the soil's nutrient depletion rate. Biochar also maintains a maximum amount of C in the soil because of its stability against microbial decay (Krull et al. 2002).
In a study by Lehmann et al. (2009) the positive effects that biochar has on the soil ecosystem’s plant and animal (microbial) life, were proposed to be as a result of the direct derivation of the nutrients found within biochar; or indirectly from biochar’s ability to sorb and retain nutrients. When biochar is added to a soil, the nutrients that biochar provides will vary depending on factors such as, and not limited to; soil pH, soil type or the type material that produced the biochar (Marks et al. 2014). All these factors have been observed to impact increase or decrease in plant available nutrients in soils amended with biochar as well as whether biochar inhibits or benefits plant growth (Schulz et al. 2009). Biochar may influence phosphorus (P) availability through various mechanisms that either directly or indirectly influence the biotic or abiotic components of the phosphorus cycle (Blackwell et al. 2009). Influence of biochar can be through bio-char acting as a modifier of soil pH ameliorating the P complexing metals such as Al3+ or Ca2+; biochar providing a direct source of soluble P salts and exchangeable P; and also as a microbial activity and P mineralization promoter (Lehmann et al. 2011).
In the case of soluble P and exchangeable P, biochar enhances P availability by releasing P salts from woody tissues during the charring process; inducing changes in the soil ion exchange capacity; sorption of plant and microbial chelates and interfering with P sorption to Al and Fe oxides (Chintala et al. 2013). Furthermore, charring cleaves organic P bonds that results in the release of soluble P salts leading to an increase in extractable PO43- as does charring biochars at tem-peratures between 350°C and 800°C (DeLuca et al. 2006).
Indirect effects of biochar on P availability can also be through provision of suitable environments for microbial organisms. These organisms recycle and produce labile pools of organic P; provide access to P from both organic and insoluble inorganic pools and can improve the direct access to P for plants through mycorrhizae (Chintala et al. 2013).
P availability is highly pH dependent. Biochar has been known to affect pH in some studies and research has concluded that there exists a possibility that biochar can modify the availability of P (Ch’ng et al. 2014). These pH changes can be related to the liming of biochar potential that is due to alkalinity with regard to either the addition of alkaline metals or the surface functional groups of biochar (Amonette and Joseph 2009). In recent years, most research studies have concentrated on the effect of biochar in acidic soils and less on soils of a calcareous nature that generally have high pH, (Lehmann et al. 2009) because of its ability to act as a liming material. It is equally importance to consider soils of a high pH, with regard to phosphorus, especially for temperate regions because generally limited understanding of the influence of biochar enhancements on fertility of soils of these regions exists (Lentz and Ippolito 2012). This study seeks to bridge that gap of limited knowledge. Calcareous soils are known to be problematic just as much as acidic soils but research under these conditions is limited. Whereas in acidic soils phosphorus might be lacking due to high acidity, depletion or inherent low soil available P; calcareous soils bind phosphates making them unavailable (Al-Rohily et al. 2013). In any case, the right level of soil acidity favors the effectiveness of phosphate fertilizers as opposed to higher pH conditions. The effects of soil acidity is reiterated in the study that shows that higher solution pH values and higher Ca:P ratios increase the pre-cipitation of phosphate to less soluble forms (Marks et al. 2014).
This study’s objectives were (1) assess the effect of biochar on P availability in either fused super phosphate (inorganic) or bone meal (organic) fertilizer amended soils (2) effect of incubation time on available P.
2. Materials and Methods
Routine analysis of the tested soil was determined according to the standard methods published by (Klute and Albert 1996; Richards 1954; Jackson 1958) and results are depicted in Table 1.
Table 1. Basic parameters of selected physical and chemical properties of pre-incubated soil, Fertilizer and Biochar
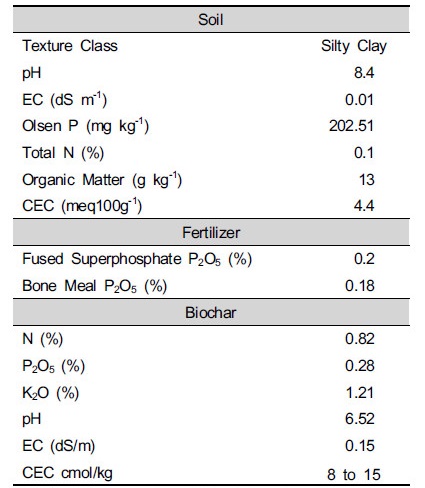 | |
*Biochar characteristics obtained from http://kd-agro.com | |
Soil for experiment collected in Siu-ri, Joan-myeon, Namyangju-si, Gyeonggi-do, Republic of Korea. Low temperature biochar sold by KD agro (http:// kd-agro.com) was selected.
Texture was determined through the pipette method while soil pH and electrical conductivity were measured in deionized water (1:5 soil to solution ratio). The total soil organic carbon was quantified by the Walkley-Black method (Yeomans and Bremner 1998) and total nitrogen was determined by Kjeldahl distillation (Bremner 1996). Phosphorus was determined colorimetrically in all extracts using the ammonium molybdate-ascorbic method (Olsen et al. 1954) and Cation Exchange Capacity (CEC) was measured with ammonium acetate (1 mol L-1) leaching method (Metson 1961).
Incubation of the soil samples; biochar amended soil (2 g biochar/100 g soil) and non-biochar amended soil enriched with either inorganic or organic fertilizer at 25 oC. Both the biochar amended soil (2%) as well as soil without biochar was divided into six samples consisting of 100g of 2 mm air dried soil sieved and weighted. The samples were; soil (control) (C), soil + biochar (SB), Soil + biochar + 40 mg P kg-1 fused super phosphate (SBF40), Soil + biochar + 80 mg P kg-1 fused super phosphate (SBF80), Soil + biochar + 120 mg P kg-1 fused super phosphate (SBF80), Soil + biochar + 40 mg P kg-1 bone meal (SBB40), Soil + biochar + 80 mg P kg-1 bone meal (SBB80), Soil + biochar + 120 mg P kg-1 bone meal (SBB120), soil + 40 mg P kg-1 fused super phosphate (SF80), soil + 40 mg P kg-1 fused super phosphate (SF80), soil + 120 mg P kg-1 fused super phosphate (SF120), soil + 40 mg P kg-1 bone meal (SBM40), soil + 80 mg P kg-1 bone meal (SBM80) and soil + 120 mg P kg-1 bone meal (SBM120). Aside from the control and SB soil, which consisted of 3 samples, the rest were amended with either inorganic or organic phosphorus fertilizer in the dose of 40, 80 and 120 mg P kg-1 in triplicate. The samples were moistened at 70% Field Water Capacity (FWC) and incubated in the dark at 25°C.
3. Results and Analysis
3.1 Effect of Time on Available P
After 70 days incubation, the effect of time of incubated treatment samples on available P is shown in Fig. 1. During the first 14 days, organic and inorganic P fertilizer containing samples had their available P significantly (p<0.005) decrease with increase in days of incubation. Decrease of available P can be generated by P fixation by soil as outlined in the study by Al-Rohily et al. (2013) whose study stated that the amount of P retained can be increased when the contact time between the soil and the phosphorus supplied increases. Overall, Inorganic P fertilizers yielded the highest amount of available P compared with organic fertilizers. The amount of extractable available P from organic sources amended with biochar was higher compared to that of soils with only organic fertilizer.
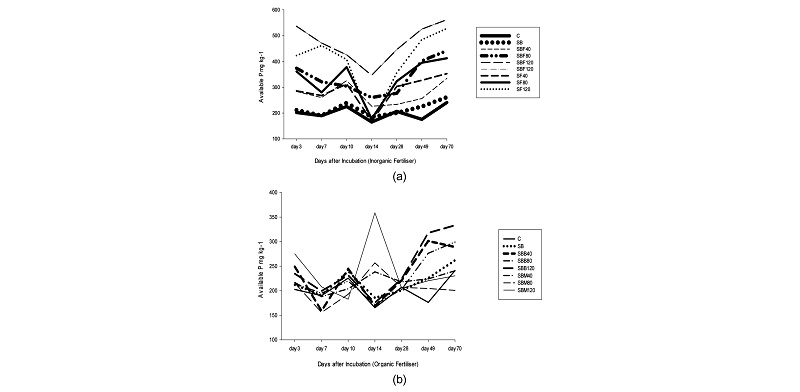
Fig. 1.
Effect of Incubation Time on Available P mg kg-1 for all inorganic (a) and organic (b) treatments. C-control, SB- soil + biochar, SBF- soil + biochar + fused super-phosphate, SBB- soil + biochar + bone meal, SF – soil + fused superphosphate, + SBM- soil + bone meal. Corresponding numeric numbers depict fertilizer dose in mg P kg-1 (P in the form P2O5).
After 14 days, samples with organic and inorganic fertilizers amended with biochar recorded an increase in available P. The results for SBB and SBM agree with the studies of Nafiu (2009), whereby samples with organic P sources show an increase in extractable available P with increase in the days of incubation. However it is important to note that organic fertilizer samples with biochar recorded an increase in available P after 14 days while those with only bone meal increased P by the 14th day then decreased only to start increasing after 28 days. The phenomenon could not entirely be understood but according to Delgado (2002) change in the composition of the soil solution by adding fertilizer to the soil can affect the dynamics of phosphorus availability.
On the contrary, the results of SBF and SF agree with previous studies only until the 14th day. These treatment sample results disagree with those of Al- Rohily (2013) and Schultz (2012) regarding inorganic fertilizer effect on available P with incubation time. Both studies found that with increasing time of incubation, inorganic fertilizer significantly decreased while that of organic fertilization increased. In the present study, the increase in available P is evident and with inorganic fertilization therefore concluding that time had a significant effect on available P in samples with inorganic fertilizer. The results agree with Naima (2015) who reported that the amount of P available from an inorganic fertilizer (Single Super Phosphate (SSP) in that study) remained significantly higher than those of other amendments and control during the final six weeks period of a study indicating that SSP under an alkaline soil conditions maintained P availability in soil for a longer period of time. The results is because the main compound in SSP (CaHPO4.2H2O) is more soluble than other Ca-P compounds that are usually formed under calcium carbonate rich soils (Hansel et al. 2014). In this case, the FSP reacted just like the SSP and SSP fertilizers under alkaline conditions present greater efficiency compared to other fertilizers. Overall, the results carried through ANOVA indicate that the effect of time in terms of days of incubation had a significant effect on the available P (p<0.001).
3.2 Effect of Fertilizers on Available P
Results of Fig. 2. show that compared to treatment samples amended with organic fertilizer (bone meal) i.e., SBB and SBM, samples containing inorganic fertilizer (FSP) yielded more extractable P. For SBF, available P across the treatment samples averages 274.51 to 473.21 mg P kg-1 while SF averages 288.45 to 403.04 mg P kg-1. Compared to SBF, from organic fertilizer, SBB averages 233.16 to 245.05 mg P kg-1 while SBM averages 218.44 to 240.03 mg P kg-1. Such divergence between FSP and BM samples can be explained by lower readily plant-available phosphorus content of BM than mineral phosphate fertilizers (Naima 2015).
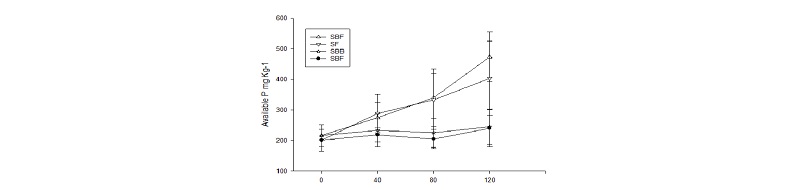
Fig. 2.
Fertilizer application rate with and without biochar for both inorganic and organic amendments. Error bars show the standard deviation for means of data calculated from averages of sample treatments by fertilization group over the incubation period. SBF- soil + biochar + fused superphosphate, SBB- soil + biochar + bone meal, SF – soil + fused superphosphate, + SBM- soil + bone meal.
Overall effect of fertilizer results shows that adding P fertilizers caused the enrichment of soil available phosphorus and that the values of inorganic fertilized samples is higher compared to the control and organic fertilizer samples, therefore agreeing with the study of Bridle and Pritchard (2004). The results with organic amendments did not show any significant difference from the controls and The results can be attributed to the short time the experiment was conducted therefore not being sufficient for the bone meal to have any significant effect on P availability as well as the issue of soil pH being high (Jeng et al. 2007).
Results show that compared to all control, all levels of treatment under FSP i.e. SBF and SF, displayed a significant effect on available P. In samples SBF120 and SF120, available P amount were showed a significant difference compared to all other samples and recorded the highest amount, 473.21 and 403.04 mg P kg-1 respectively. In comparison, results from treatment samples containing bone meal with biochar were not significantly different from neither those of control nor with only bone meal amendment. The only sample that recorded a significant difference from control was SBB120. Samples containing 80 mg kg-1 for inorganic fertilization with and without biochar, SBF80 and SF80 recorded means that were not significantly different and the same applied for samples SBF40 and SF40. Furthermore, samples containing 80 mg kg-1 recorded even lower amount of available P in comparison to samples containing 40 mg kg-1. Results show that SBB40 and SBM40 had 233.16 and 218.44 mg P kg-1 respectively compared to SBB80 and SBM80, 225.39 and 204.64 mg P kg-1.
These results disagree with results from Nogalska (2013), Al-Rohily et al (2013) and Shultz, (2012) whereby the increase in available P with organic amendment increased in proportion to the amendment application rate. Delgado (2002)’s study that the change in the composition of the soil solution by adding fertilizer to the soil can affect the dynamics of phosphorus can be applied here if one was to try and understand the reason why 80 mg kg-1 samples were lower than 40 mg kg-1 organic samples.
4. Discussion
Researchers advise that before considering biochar’s influence on nutrient transformations, that the nutrient capital associated with biochar additions be considered because the obvious attribute of adding biochar is its nutrient value (Chan 2009). The value is supplied either directly or indirectly for instance by improvement of soil quality with the subsequent improvement of fertilizer use.
However, as this study has shown, that is not always the case. Incubated samples containing both inorganic and organic fertilizer with and without biochar all significantly increased P (p<0.05) than control samples. However, addition of biochar or the lack of it did not significantly impact available P when samples containing fertilizers with biochar were compared to those without biochar. Study by Keeney (1982) Bridle and Pritchard (2004) stated that with biochar, the total content of nutrients is not an appropriate indicator of the availability of nutrients. Only a fraction of the total content is readily/ immediately available for plant uptake and for organically bound nutrients, their total elemental contents do not necessarily reflect their actual avail-ability (Chan 2009). We can therefore assume that despite the biochar used in this study having a substantial amount of total P, under the experimental conditions here it was not necessarily available, other factors considered as well, to necessarily have a significant effect on increasing available P either used with fertilizers or not.
Another study by Schultz (2012) with biochar that did not yield any effect on nutrient availability was attributed to the amount of biochar application rate as a possible reason. In that particular study, Schultz concludes that the no effect from biochar was that compared to other biochar success studies his study may have utilized minimal additions of biochar in comparison to higher biochar application amount. And the study also explained that using compost and fertilizer with such minimal amount of biochar could mask the biochar effects and therefore not show any significant effect on the availability of nutrients using the amendment. With the same reasoning, the current study’s application of 2% w/w biochar application may have been overpowered by the effects of both the organic and inorganic fertilizer effects on availability of Schultz (2012) further explained that in cases where biochar is co-used with fertilizer or organic amendment, it would be deemed effective if it is applied in agronomical higher amonts than when used without fertilizers.
The purpose of the present study was to determine the influence of biochar with phosphorus fertilizers applied on the soil and its impacts on the availability of phosphorus during a short-term incubation ex-periment. These biochar with phosphorus amendments were performed at lab scale, in closed conditions. Thus, they might not be in complete agreement with the natural phenomenon occurring outside of the lab. However, they are performed to gain more insight into the interactions of biochar and phosphorus fertilizers in determining phosphorus availability.
In conclusion, overall results show that time was a significant factor in P availability with incubation. Results for inorganic fertilizer recorded the highest amount of available P and for a longer period. However more time is required to observe the outcome of trend. Pertaining to biochar, it did not have any significant effect on the availability of phosphorus with inorganic fertilization while the time period was too short and soil conditions unfavorable to discern the effect of bone meal and bone meal with biochar on the availability of P. It is recommended that the experiment be carried for a longer time period to observe the possible contributions that bone meal with biochar can have on P availability similar with the soil pH.
Concerning inorganic fertilization an extended time period would also be effective in observing if the samples with biochar, especially 40 and 80 mg P kg-1 can surpass their counterparts without biochar amend-ment.




